 Detailed
introduction
Detailed
introduction
1. Features and Scope of Application
Some catalysts used in hydrocarbon-based large-scale
ammonia plants is sensitive to sulfur compounds and prone to be poisoned and
deterioration in activity when sulfur content in feed gas exceeds certain
value. Cobalt –molybdenum hydroconversion catalyst and zinc oxide are usually
used for desulfurization of feed gases or oils.
T201 hydroconversion catalyst, with high organic sulfur
conversion capability, is applicable to hydroconversion of feed gases for
large-scale ammonia plants. It can bring down organic sulfur in feed gases to
less than 0.1 ppm.
Main hydroconversion reactions involved are as follows:
RSH+H2 = RH+H2S
R1SSR2+3H2 =
R1H+R2H+H2S
R1SR2+2H2 =
R1H+R2H+H2S
C4H4S+4H2 =
C4H10+H2S
COS+H2 = CO+H2S
where R=alkyl groups.
This product is also applicable to organic sulfur
hydroconversion of light oils or gaseous hydrocarbons in petrochemistry.
2. Physical properties

3.Quality norm
According to HG2505-93 industrial standard, catalyst
T201should conform to following norm:

4. Reference operating conditions

The hydrogenation reactions take place at 300-450℃.
Initial temperature is usually controlled at 350-380℃. If
sulfur concentration in feed oil stays below a certain limit ( i.g. , 0.2ppm)
for long term, “Sulfur discharge” phenomenon will take place. Therefore, for
two-section oil hydrogenation system, operating temperature in 1st section should be such that ensures sulfur concentration of 2—10ppm in effluent
naphtha , so as to maintain the catalyst in 2nd section in sulfided
state.
5. Loading
(1)Clean the reactor of any debris and screen the
catalyst of any powder before loading. Operators working inside the reactor
should stand on a broad wooden plate without stepping directly on the catalyst.
(2)Install inert balls at the top and bottom of the
reactor . The catalyst particles are separated from the inert balls by
stainless wire net of smaller mesh size than the catalyst.
(3)Use a funnel connected by a S-type cloth tube to drop
the catalyst slowly and evenly from a maximum height of 1.2m to the reactor
while holding lower end of the tube to prevent breakage of the particles.
(4)The loading operators should not stand directly on the
catalyst bed during loading.
6. Start-up and Catalyst Presulfiding
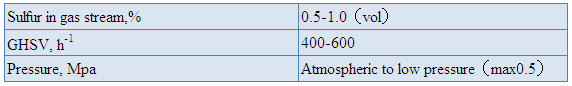
Purge the system with nitrogen or other gases and then
warm up the catalyst bed with nitrogen, hydrogen-nitrogen or natural gas.
Warming-up procedure: 30~50℃/h to
120℃,
keep at 120℃
for 2 h, and then 30~50℃/h to
220℃.
Then carry out presulfiding while warming up.
Usually presulfiding is not necessary for first-time use
of the catalyst when using natural gas, associated gas or light naphtha as
feedstock, since inorganic sulfur in the gaseous feed may fulfill sulfiding
gradually during operation. However, in the case of treating hydrocarbons with
high and/or complicated sulfur , presulfiding is need for first-time use to
attain higher hydrogenation activity. Sulfur absorbed amounts to about 5% of
total weight of the catalyst at the end of presulfiding.
Presulfiding can be done in following two ways:
(1)Adding CS2 into nitrogen or hydrogen
Add CS2 into feed gas (hydrogen-nitrogen or
hydrogen) after warming up to 220℃. Carry out presulfiding while warming up at
20℃/h
till operating temperature. Presulfiding can be considered complete when
sulfur-containing gas equivalent to theoretical sulfur adsorbent capacity of
the catalyst is added.
Presulfiding condition:
2)Adding CS2 into light oil(preferably
light naphtha)
Pass sulfiding medium into catalyst bed when bed
temperature reaches 220℃. Keep on sulfiding while warming up at 20℃/h till
operating temperature. Presulfiding can be considered complete when sulfiding
medium equivalent to theoretical sulfur absorption capacity of the catalyst is
added. Then raise pressure to operation condition, shift to hydrocarbon feed
and adjust temperature, LHSV and hydrogen/oil, and and gradually proceed to
normal full load operation.
Properly enhance operating temperature in later service
stage of the catalyst to increase its activit.
Presulfiding condition:

7 Shutdown
(1) Temporary shutdown
For liquid feed, stop feed supply , purge the system for
1 h to remove any liquid hydrocarbon, close inlet and outlet valves and
maintain temperature and pressure in the reactor. For gaseous feed, cut feed
supply and maintain the pressure.
(2)Long-term shutdown
For long-term shutdown without disassembly of the
reactor, lower to 30% load, lower the temperature at 30-50℃/h to
250℃
and the pressure to 1.5MPa at no greater than 0.5MPa/h to avoid catalyst
breakage. Then stop supply of feed , purge the system with hydrogen for 1 h,
close inlet and outlet valves, maintain the pressure at positive (no less than
0.1MPa) and let the temperature drop naturally. For gaseous feed, Cut feed
supply and bring down the pressure and temperature at above-mentioned rate.
For long-term shutdown with disassembly of the reactor,
purge the system with nitrogen, maintain positive pressure and bring down
temperature to 40℃ disassembly.
(3)Start-up after shutdown
The same procedure as initial start-up. For liquid feed,
to avoid reduction of the catalyst (especially over 250℃), warm
up with nitrogen or inert gas until operating temperature. Then shift to feed
oil and hydrogen. For gaseous feed, warm up directly with gaseous feed and
hydrogen.
When hydrogenating gas is used for the warming-up, pass
hydrocarbon feed into the reactor immediately after the temperature exceeds dew
point of the liquid hydrocarbon, and then continue warming-up till operating
temperature.
(4)Incidental shutdown
Due to variety of causes of incidents, no general-purpose
procedure can be given out for incidental shutdown. Following are tips to be
paid attention to for avoiding harm to the catalyst:
1 Lowering temperature at over 50℃/h when
reactor temperature is higher than 200℃ is harmful to both strength and activity and
service life of the reactor.
2 The reactor can tolerate short time interruption of
hydrogen supply (several minutes). Long time interruption may cause coke
formation on the catalyst, sometimes so serious that regeneration or changeover
is necessary.
3 Long-term
contact with sulfur-free hydrogen at over 250℃ may cause reduction and hence activity loss
of the catalyst.
8. Regeneration
Activity of the catalyst may deteriorate with service
time due to coke formation. When this deterioration becomes intolerable to
operation requirement, it is necessary to regenerate the catalyst.
Shut down according to the procedure for “long-term
shutdown without disassembly” . Bring down temperature to 250℃ and
pressure to atmospheric and then pass air-containing steam ( 0.5-1.0% oxygen)
into reactor for regeneration. Increase oxygen concentration in the steam with
temperature rise till totally air . Maintain at 450℃
(maximum 475℃)
for 4h after there is no temperature rise and oxygen concentration at the inlet
and outlet becomes equal. Then regeneration can be considered completed.
When rapid temperature rise is observed while increasing
oxygen concentration in steam, stop adding air and pass solely steam to relief
temperature rise. Resume and increase addition of air when temperature becomes
normal. Exothermal reaction may take place and cause remarkable temperature
rise at 350-400℃.
Strictly control air addition and prevent damage to the catalyst by temperature
surge.
Analysis of oxygen and CO2 concentration in
outlet stream is helpful to inspect progress of regeneration. Regeneration can
be considered completed when oxygen in inlet and outlet stream approaches the
same. Continue to pass air flow and bring down temperature at 40~50℃/h to 220℃. Then shift to nitrogen purging and
presulfiding and finally normal operation.
Regeneration cycle is 2-3 years under normal operation
condition.
9. Package and Storage
The catalyst is packed in iron barrel lined inside with
plastic bags. It should be stored in dry and cool place. The catalyst usually
can be stored for several years without remarkable deterioration in properties
and activity.
During the regeneration to prevent temperature surge
which may cause loss of activity of the catalyst.